
With mRNA vaccine technology reshaping our approach to disease prevention, it opens up a world of possibilities in the realm of healthcare. This innovative technology not only addresses current infectious diseases like COVID-19 but also holds promise for future applications in cancer treatment and beyond. As we delve into this fascinating subject, we uncover how mRNA vaccines are designed, their efficacy compared to traditional vaccines, and the impact they could have on dentistry and medicine.
The foundational principles of mRNA vaccine technology involve using messenger RNA to instruct cells to produce a protein that triggers an immune response, offering a faster and potentially more effective way to combat diseases. The development process, from conception through clinical trials to regulatory approval, showcases the remarkable advancements in scientific research that have made this technology a cornerstone in modern medicine.
Overview of mRNA Vaccine Technology
mRNA vaccine technology represents a groundbreaking approach in the development of vaccines, leveraging the body’s own cellular machinery to elicit a robust immune response. This innovative method has gained significant attention, especially in the context of infectious diseases, providing a platform for rapid response to emerging pathogens.The fundamental principle behind mRNA vaccines involves the use of messenger RNA (mRNA) to instruct cells to produce a harmless piece of the target pathogen, usually a protein.
This protein acts as an antigen, triggering the immune system to recognize and mount a defense against the actual pathogen if encountered in the future. Unlike traditional vaccines, which often use weakened or inactivated forms of the virus, mRNA vaccines utilize synthetic mRNA that encodes the relevant protein, leading to a more efficient production process and faster vaccine development times.
Diseases Targeted by mRNA Vaccines
Several diseases are now targeted by mRNA vaccines, showcasing their versatility and effectiveness. Notably, mRNA technology has made significant contributions to the fight against the following diseases:
- COVID-19: The most prominent example is the Pfizer-BioNTech and Moderna vaccines, which have been pivotal in combating the COVID-19 pandemic. They encode the spike protein of the SARS-CoV-2 virus, stimulating an immune response that helps prevent severe illness.
- Influenza: Research is ongoing to develop mRNA vaccines for seasonal flu, aiming to enhance efficacy and adaptability compared to traditional flu vaccines.
- Zika Virus: mRNA vaccine candidates have shown promise in preclinical trials, offering a potential strategy to combat this mosquito-borne virus.
- Rabies: mRNA vaccines are being explored as a novel approach to rabies vaccination, which may simplify immunization efforts in at-risk populations.
Development Process of mRNA Vaccines
The development of mRNA vaccines follows a systematic process, from initial conception to regulatory approval. This journey can be Artikeld in several key stages:
1. Research and Design
Once a target pathogen is identified, researchers design the mRNA sequence that encodes the antigen of interest. Computational tools are often employed to optimize the sequence for efficient translation and immune recognition.
2. Preclinical Testing
Before moving on to human trials, mRNA formulations undergo rigorous testing in laboratory settings and animal models to assess safety and immunogenicity. This stage is crucial for identifying potential side effects and determining the optimal dosing regimen.
3. Clinical Trials
If preclinical results are promising, the vaccine progresses to clinical trials, typically divided into three phases:
Phase 1 focuses on safety, assessing the immune response in a small group of healthy volunteers.
Phase 2 expands the participant pool to evaluate efficacy and further assess safety among different demographics.
Phase 3 involves thousands of participants to confirm efficacy and monitor for adverse effects, ultimately leading to the collection of comprehensive data.
4. Regulatory Approval
After successful clinical trials, vaccine developers submit their findings to regulatory agencies such as the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). This includes a thorough review of all data to ensure that the vaccine meets safety and efficacy standards.
5. Manufacturing and Distribution
Once approved, large-scale manufacturing begins, utilizing robust quality control measures. The final product is then distributed to healthcare providers and vaccination sites.This structured development process highlights the meticulous care taken to ensure that mRNA vaccines are both safe and effective before being administered to the public.
Applications of mRNA Vaccine Technology in Healthcare

The versatility of mRNA vaccine technology has positioned it as a pivotal player in modern healthcare, particularly in the realms of infectious diseases and cancer treatment. The rapid development of mRNA vaccines during the COVID-19 pandemic has showcased not only their efficacy but also their potential applications across a range of diseases. This technology enables the body to produce proteins that provoke an immune response, which opens up numerous avenues for therapeutic interventions and preventative measures.
Potential of mRNA Vaccines in Combating Emerging Infectious Diseases
mRNA vaccines represent a revolutionary approach in the fight against emerging infectious diseases. Their ability to be rapidly developed and deployed allows for swift responses to newly identified pathogens. The technology is particularly valuable in situations where traditional vaccine development could take years or even decades. Recent examples include:
- COVID-19: The unprecedented speed of mRNA vaccine development against SARS-CoV-2 demonstrated how quickly these can be adapted to new variants, providing effective protection in record time.
- Zika Virus: Research is ongoing to evaluate mRNA vaccines targeting the Zika virus, which poses significant risks during pregnancy, showing promise in generating a robust immune response.
- Influenza: mRNA technology is being explored to create seasonal flu vaccines that can be updated more efficiently than traditional methods, potentially leading to broader and more effective immunization strategies.
The adaptability of mRNA platforms ensures they remain at the forefront in combating other infectious agents like HIV and Ebola, where traditional vaccines have struggled to elicit strong and lasting immune responses.
Role of mRNA Vaccines in Cancer Immunotherapy
The application of mRNA technology in cancer immunotherapy is gaining significant traction, as it offers a personalized approach to treatment. mRNA vaccines can be designed to encode specific tumor-associated antigens, prompting the immune system to recognize and attack cancer cells.Important aspects of this application include:
- Personalized Cancer Vaccines: mRNA vaccines can be tailored to individual patients by encoding unique neoantigens derived from their tumors, enhancing the specificity and efficacy of the immune response.
- Combination Therapies: mRNA vaccines are being investigated in combination with checkpoint inhibitors and other immunotherapies to improve overall treatment outcomes.
- Clinical Trials: Numerous clinical trials are underway assessing the safety and efficacy of mRNA vaccines in various cancer types, including melanoma and breast cancer, demonstrating promising early results.
The potential for mRNA vaccines to provide targeted and effective treatments exemplifies a paradigm shift in how cancer therapies are developed and administered.
Comparison of Traditional Vaccines and mRNA Vaccines
A comparison of mRNA vaccines with traditional vaccines reveals key differences in efficacy and safety profiles. Understanding these differences is crucial in appreciating the advancements offered by mRNA technology.
| Feature | Traditional Vaccines | mRNA Vaccines |
|---|---|---|
| Development Time | Longer, often years | Rapid, can be developed in months |
| Efficacy | Varies widely | High efficacy demonstrated (e.g., COVID-19) |
| Storage Requirements | Stable at standard refrigeration | Requires ultra-cold storage |
| Immune Response | May require multiple doses | Strong responses, often with fewer doses |
| Production | Requires growing live pathogens | Manufactured using synthetic processes |
| Safety Profile | Established but can vary | Generally favorable, with ongoing monitoring |
This comparison highlights the innovative nature of mRNA vaccines, showcasing not only their efficiency but also the potential challenges they present in terms of storage and production. The shift from traditional methods to mRNA technology marks a significant evolution in vaccine development and public health strategies.
Impact on Dentistry and Medicine
The advent of mRNA technology has opened new avenues not only in vaccine development but also in the broader fields of dentistry and medicine. This innovative approach holds promise for enhancing patient care, targeting specific diseases, and potentially revolutionizing treatment protocols. By harnessing the power of mRNA, healthcare professionals can aim for more personalized and effective therapies that address a range of health concerns, including oral diseases.mRNA technology presents a transformative potential in the development of dental vaccines aimed at preventing common oral diseases.
Unlike traditional vaccines, which often use weakened or inactivated pathogens, mRNA vaccines instruct cells to produce proteins that elicit an immune response. This method could be particularly beneficial in targeting periodontal disease and dental caries, where the immune system’s response plays a crucial role. By creating specific mRNA sequences that encode for antigens associated with these diseases, researchers can develop vaccines that not only prevent disease onset but also enhance the body’s ability to combat existing infections.
Ongoing Research in mRNA Vaccines for Oral Diseases
Research into mRNA vaccines for oral diseases is in its nascent stages, yet several promising studies are underway. Investigations are focusing on the efficacy of mRNA platforms in addressing pathogens responsible for oral infections. The following points summarize key areas of ongoing research:
- Periodontal Disease: Research teams are exploring mRNA vaccines that target specific bacteria implicated in periodontal disease, aiming to provoke a robust immune response that could reduce inflammation and tissue destruction.
- Dental Caries: Ongoing studies are assessing mRNA vaccines designed to promote immunity against the bacteria Streptococcus mutans, a primary contributor to tooth decay, potentially offering a preventive measure for caries development.
- Oral Cancer: Innovative projects are looking into mRNA-based therapies that encode cancer-specific antigens, potentially leading to personalized vaccines for patients with oral squamous cell carcinoma.
The exploration of these areas highlights the significance of mRNA technology in dental health and its potential to introduce novel treatment modalities that could significantly impact patient outcomes.
Framework for Integrating mRNA Technology in Future Medical and Dental Practices
As mRNA technology continues to evolve, integrating it into routine medical and dental practices requires a comprehensive framework. This framework should encompass several key components to ensure effective implementation and patient safety:
1. Collaboration Across Disciplines
It is essential for dental professionals, researchers, and healthcare policymakers to work together. By fostering interdisciplinary collaborations, insights from varied fields can address challenges in vaccine development and deployment.
2. Clinical Trials and Regulatory Pathways
Establishing clear protocols for clinical trials is critical in evaluating the safety and efficacy of mRNA vaccines. Adhering to regulatory requirements will be necessary to gain approval for clinical use.
3. Patient Education and Engagement
Informing patients about the benefits and risks associated with mRNA vaccines is vital. Engaging patients in discussions can enhance their understanding and acceptance of new therapies.
4. Continued Research and Development
Ongoing investment in research is crucial to explore various applications of mRNA technology. This includes refining vaccine formulations and delivery methods to maximize efficacy.
5. Monitoring and Evaluation
Post-implementation, continuous monitoring of mRNA vaccine outcomes will provide valuable data on efficacy and safety, guiding further innovations and adjustments in treatment protocols.By implementing this framework, the integration of mRNA technology into medical and dental practices can be seamless and patient-centered, paving the way for advanced therapeutic options in the future.
Last Point
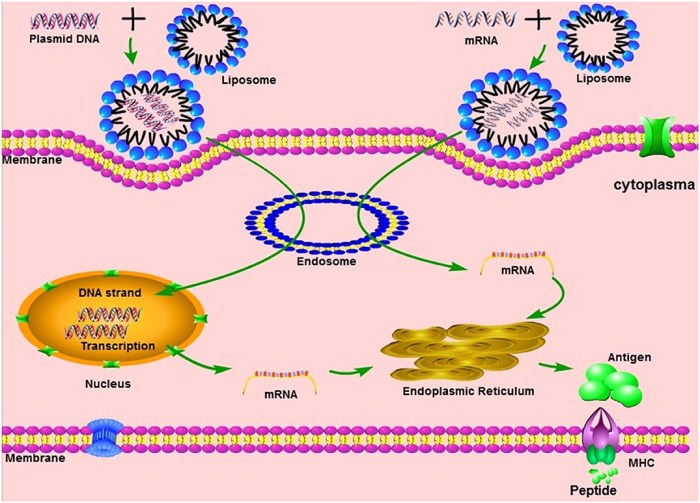
In summary, mRNA vaccine technology represents a significant leap forward in our ability to prevent and treat diseases, highlighting the potential for innovative healthcare solutions. Its ability to adapt to emerging threats and its applications in areas like cancer immunotherapy and dental health reflect a promising future. As researchers continue to explore the full scope of mRNA technology, we can anticipate a transformative impact on both individual health and public health on a broader scale.
FAQ Compilation
What is mRNA technology?
mRNA technology uses messenger RNA to instruct cells to produce proteins that elicit an immune response, offering a novel approach to vaccination.
Are mRNA vaccines safe?
Yes, mRNA vaccines have undergone rigorous testing in clinical trials to ensure their safety and efficacy before receiving emergency use authorization or full approval.
Can mRNA vaccines be used against cancer?
Yes, ongoing research is exploring the use of mRNA vaccines in cancer treatment by helping the immune system recognize and attack cancer cells.
How do mRNA vaccines compare to traditional vaccines?
mRNA vaccines typically have a faster development time and can be more easily adapted to target new pathogens compared to traditional vaccines.
What diseases are currently targeted by mRNA vaccines?
Currently, mRNA vaccines are used primarily for infectious diseases like COVID-19, but research is expanding into several other diseases, including influenza and various cancers.





